Hyun-Joon Ha
Hankuk University of Foreign Studies, Korea
Title: Polyhydroxy alkaloids from aziridine
Biography
Biography: Hyun-Joon Ha
Abstract
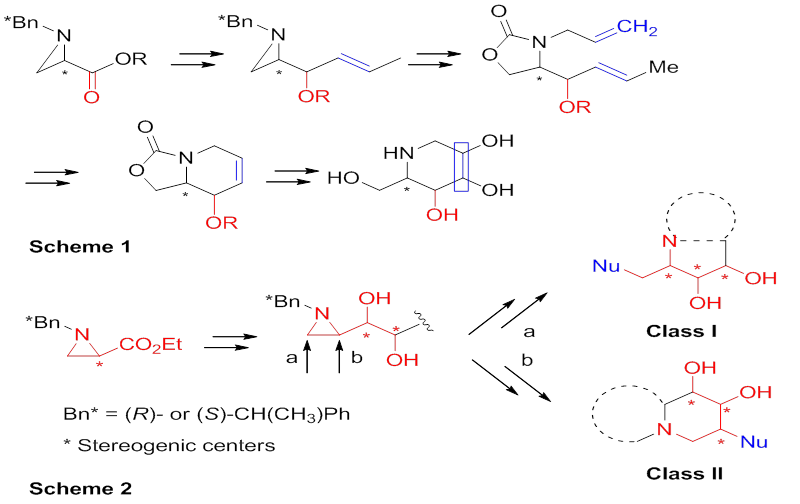
Alkaloids are nitrogen-containing natural products found in diverse organisms including fungi, plants, and animals. Among them, polyhydroxy alkaloids have a plethora of intriguing biological activities. Owing to their importance within the realm of medicinal and bioorganic chemistry, there is a rich literature detailing the attempts towards the synthesis of natural polyhydroxy alkaloids and their analogs. In most cases, those were prepared from uniquely functionalized starting materials with the assistance of a series of chemical transformations involving the construction of the carbon backbones followed by the stereoselective installation of hydroxyl groups. These synthetic strategies do not allow preparing multiple synthetic targets from a common synthetic intermediate. A chiral aziridine-2-carboxylate is a good starting material to elaborate polyhydroxy alkaloids, many of which were prepared in our laboratory in highly efficient manner with structural diversities. One of the typical examples is 1-deoxynojirimycin derived from hydroxyl functionalities using the common advanced synthetic intermediate via different hydroxylation methods. In additional polyhydroxy pyrrolidine alkaloids were also synthesized by branching two possible regiochemical pathways of ring-opening reactions of 2-substituted aziridine as a common synthetic intermediate providing an easy access to two different classes of compounds. Efficient regiochemistry-directed syntheses of monocyclic, bicyclic, and acyclic polyhydroxy alkaloids were achieved by taking different regiochemical pathways of aziridine ring-opening reactions of dihydroxylated aziridine as a common synthetic intermediate, which was readily available from commercial aziridine-2-carboxylate. Two possible regiochemical pathways of aziridine ring opening provided an easy access to two different classes of compounds from a common synthetic intermediate, called as regioselectivity-directed branches of 2-substituted aziridine. The applications of this synthetic strategy diversifing the stereoselective dihydroxylation and regioselective aziridine-ring opening as the key steps allowed the asymmetric synthesis of natural and unnatural polyhydroxy alkaloids including calyculin fragment C33-C37, 1,4-dideoxy-1,4-imino-L-ribitol and analogs of hyacinthacine, swainsonine, castanospermine and deoxynojirimycin.